Every few months we highlight a subset of the new plasmids and viral preps in the repository through our hot plasmids articles. These articles provide brief summaries of recent plasmid deposits and we hope they'll make it easier for you to find and use the plasmids you need.
 Here's what you'll find in this post:
Here's what you'll find in this post:
- Epigenetic engineering with single-pot assembly
- Tracking cell-cell interactions
- New auxin inducible degron system for C. elegans
- New CRISPR plasmids
- New items from the viral service
Epigenetic engineering with single-pot assembly
By Eric Perkins
Though it’s easy enough to contemplate DNA in terms of its sequencing, in vivo studies should always take DNA’s chromatin status into account (Fig 1a). The laboratory of Karmella Haynes at Emory recently contributed a set of plasmids to the repository that will help epigenetic researchers custom design their own engineering plasmid tools in about a week. Taking advantage of Golden Gate cloning, the Haynes lab’s Protocols.io protocol describes an elegant system that allows for the assembly of multiple DNA fragments efficiently and accurately in a “single pot.’
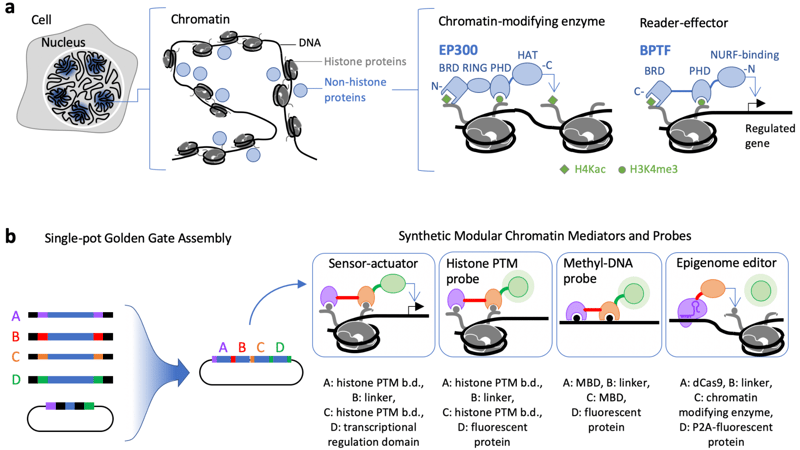 |
| Figure 1: Single-Pot Assembly: a) Graphic depiction of chromatin within a nucleus -> DNA compaction around histones -> Examples of histone-modifying enzymes and reader-effector proteins. b) Golden Gate assembly (backbones available at Addgene) with examples of potential synthetic chromatin mediators and probes. Image from Priode et al., 2021. |
The potential tools fall into two categories: probes and editors. A probe array may consist of at least two sensor modules targeting a specific chromatin feature (e.g. a methylated histone such as H3K27me3), a linker between these modules (linker length can and should be varied as it can have important implications for the fusion protein effectivity), and a fluorescent protein tag for visualization (Fig. 1b). While the probes target chromatin to visualize DNA, an editor targets DNA (via catalytically inactive dCas9) to modify chromatin. A fluorescent protein can still be included for visualization purposes, but the key component of the editing assemblies are sensor-actuators, either transcriptional regulators or chromatin-modifying enzymes. The speed and flexibility of the single-pot system will allow investigators to rapidly create multiple synthetic chromatin-modifying proteins quickly and accurately.
Priode and Haynes, protocols.io. 2021. dx.doi.org/10.17504/protocols.io.brgcm3sw
New system developed to track cell-cell interactions
GFP-based Touching Nexus (G-baToN) is a new tool developed by Rui Tang in Monte Winslow’s lab to label cells after cell-cell interactions. Using G-baToN, “sender” cells express GFP on the surface and “receiver” cells express an anti-GFP nanobody on the cell surface. When the cells interact, GFP is transferred to the receiver cell, and the receiver cell turns green. This system can be used in many cell types including endothelial cell-pericyte, neuron-astrocyte, and cancer-stromal cell pairs.
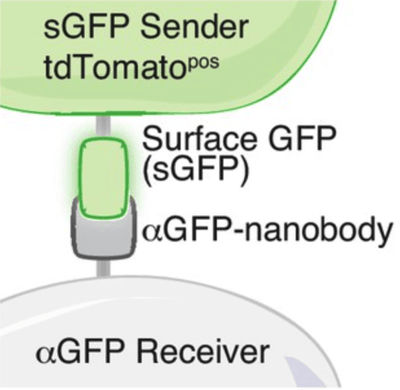 |
| Figure 2: Schematic of the interaction between the sender cell and the receiver cell. Image from Tang et al., 2020. |
G-baToN can also be applied to other fluorophore/antibody pairs. Because the anti-GFP nanobody cross-reacts with BFP, sender cells expressing surface BFP generated BFP-labeled receiver cells. What’s more, G-baToN can be used to bring cargo, such as protein or DNA, to manipulate function in receiver cells. There are over 40 trillion cells that make up the human body and G-baToN provides versibility in tracking and modulating the interactions between cells.
Tang et al., eLife 2020. https://doi.org/10.7554/eLife.61080
A new tool-kit for the auxin inducible degron (AID) system in C. elegans
The auxin inducible degron (AID) system has been extensively used in C. elegans since 2015 to conditionally degrade proteins of interest in cells (Zhang et al., 2015). Recently the Ward lab created a tool-kit to expand upon these original tools that allows for both spatiotemporal protein degradation and quantification of this depletion using imaging-based measurements (Ashley et al., 2021).
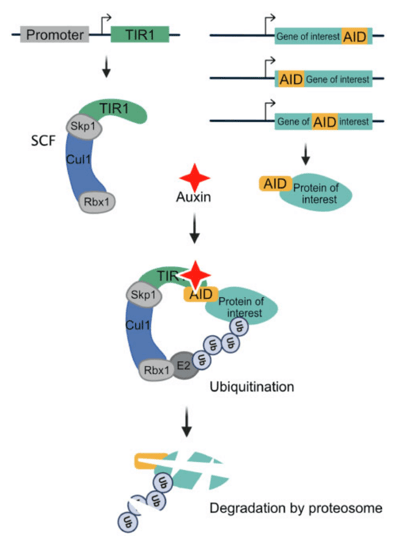 |
| Schematic showing the auxin-inducible degron system. Image from Ashley et al., 2021. |
The AID system relies on a transport inhibitor response 1 (TIR1) expressed under a desired promoter with a specific spatiotemporal expression pattern and an auxin inducible degron (AID) sequence attached to your protein of interest. TIR1 interacts with endogenous Skp1 and Cul3 proteins to form a SCF E3 ubiquitin ligase complex. In the presence of auxin, this TIR1 complex binds to the degron sequence ultimately leading to the degradation of the AID-tagged protein. To quantify protein degradation the Ward lab used fluorescent proteins (FPs) fused to the AID* (minimal 44 amino acids auxin inducible degron sequence) tagged proteins. Degradation of your protein interest would also result in depletion of the FP, which can be easily visualized.
The tool-kit consists of highly useful vectors compatible with Gibson and Saptrap cloning systems to generate AID* tagged CRISPR knock-in repair constructs. These repair constructs can be used to integrate the tagged AID* into genomic sites of interests via CRISPR techniques. This tool-kit includes an array of FPs as well as additional useful tags, including 3xFLAG epitopes, TEV and BioTag, which can be used in protein affinity purification studies.
Duong et al., Genetics 2021. https://doi.org/10.1093/genetics/iyab006
The CRISPR corner
Here are a few highlights from recent CRISPR plasmids. To find all of the CRISPR plasmids available from Addgene, head over to our CRISPR Plasmids and Resources page.
- Plasmids for delivering CRISPRi components by a single AAV vector are now available from Tonia Rex's lab.
- vfCRISPR, or very fast CRISPR, is a light-inducible CRISPR system developed by Taekjip Ha’s lab where Cas9-gRNA complex binds the target DNA without cleavage until it's activated by light. Once activated, Cas9 cleavage can induce DSBs within minutes.
- New CRISPR plasmids for Borrelia burgdorferi from Christine Jacobs-Wagner’s lab produce different levels of dCas9 expression and contain different antibiotic resistance markers.
- New CRISPR pooled library from Keith Joung’s and Benjamin Kleinstiver’s labs are now available. These libraries encode target sites flanked by randomized nucleotides in place of a protospacer adjacent motif (PAM). They're used to determine the PAM requirements of Cas enzymes.
- CRISPR-ARE is a new system from Jiazhang Lian’s lab that enables simultaneous gene activation, repression, and editing with a single Cas9-VPR in Saccharomyces cerevisiae.
- To control Cas9 activity and influence DNA repair pathways, Amit Choudhary's lab developed a chemogenetic system that brings Cas9 to ubiquitin ligase for degradation.
New from the viral service
By Jason Nasse
We regularly add new viral aliquots from our plasmid collection to provide ready-to-use viral preps. Here are some of the new viral preps from recent months:
- New INTRSECT viral preps available! We are expanding our inventory of ready to use viral preps from the Deisseroth lab INTRSECT project. Check out the new additions.
- Image calcium transients in PV interneurons with AAV-S5E2-GCaMP6f, now available as a ready-to-use viral prep in AAV1!
- New ready-to-use AAV prep from Hongkui Zeng's lab are available for dual expression of cytoplasmic-tdTomato and Synaptophysin-GFP.
- Synapse targeting GCaMP6s from the Rylan Larsen lab, now available in a ready to use AAV5 viral prep.
- Flp-dependent DREADDs from the Gether lab, now available as ready-to-use AAV vectors.
Topics: Hot Plasmids, Plasmids









Leave a Comment