Originally published Feb 6, 2014 and last updated Nov 10, 2020.
Now that we know all about antibiotic resistance genes, let’s consider another basic element of any plasmid: the origin of replication/replicon. The replicon is comprised of the origin of replication (ori) and all of its control elements. The ori is the place where DNA replication begins, enabling a plasmid to reproduce itself as it must to survive within cells.
The replicons of plasmids are generally different from the those used to replicate the host's chromosomal DNA, but they still rely on the host machinery to make additional copies. ori sequences are generally high in As and Ts. Why, you ask? Well, A-T base pairs are held together with two hydrogen bonds not three as G-C pairs are. As a result, stretches of DNA that are rich in A-T pairs can be separated more readily at lower temperatures and allows the replication machinery room to come in and get busy making copies.
Types of origins of replication
There are lots of origins of replication out there so, for simplicity’s sake, we've ignored those used in eukaryotic cells and viruses and focused only on those found in bacteria. Some common ones you might see include ColE1, pMB1 (which comes in a few slightly different but well known derivatives), pSC101, R6K, and 15A. Not all origins of replication are created equal. Some will produce many plasmid copies and others produce just a few copies depending on how they are regulated. Generally, control of replication is referred to as "relaxed" or "stringent" depending on whether the ori is positively regulated by RNA or proteins, respectively. A plasmid's copy number has to do with the balance between positive and negative regulation and can be manipulated with mutations in the replicon. For example, the pMB1 ori maintains about 20 copies per cell, while pUC – which differs by only two mutations – will produce as many as 700 copies per cell.
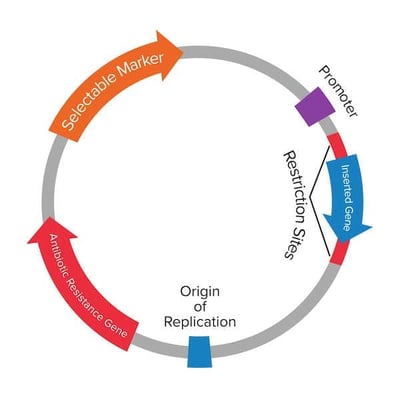 |
| Figure 1: A plasmid map showing the standard features of a plasmid. |
So, how do you choose? Addgene Senior Scientist Marcy Patrick says researchers can ask themselves a few questions to get started:
- Will the plasmid be used exclusively in E. coli? Gram negative bacteria in general? Both Gram negatives and Gram positives?
- Will you have only one plasmid type in your cells at a time?
- Do you want to make a lot of your plasmid?
- Is the gene toxic in high amounts? It is always good to keep in mind that plasmids with low to medium copy numbers can still express massive amounts of protein given the proper promoter and growth conditions.
Choose your origin of replication wisely and avoid plasmid incompatibility
In other words, the best choice of ori depends on how many plasmid copies you want to maintain, which host or hosts you intend to use, and whether or not you need to consider your plasmid's compatibility with one or more other plasmids. Generally speaking, plasmids with the same ori are incompatible because they will compete for the same machinery, creating an unstable and unpredictable environment. As a rule, plasmids from the same group should not be co-transformed, so if you need two plasmids for an experiment, make sure they have "compatible" ori's. See the table below for more details.
| Common Vectors | Copy Number+ | ori | Incompatibility Group | Control |
| pUC | ~500-700 | pMB1 (derivative) | A | Relaxed |
| pBR322 | ~15-20 | pMB1 | A | Relaxed |
| pET | ~15-20 | pBR322 | A | Relaxed |
| pGEX | ~15-20 | pBR322 | A | Relaxed |
| pColE1 | ~15-20 | ColE1 | A | Relaxed |
| pR6K | ~15-20 | R6K* | C | Stringent |
| pACYC | ~10 | p15A | B | Relaxed |
| pSC101 | ~5 | pSC101 | C | Stringent |
| pBluescript | ~300-500 | ColE1 (derivative) and F1** | A | Relaxed |
| pGEM | ~300-500 | pUC and F1** | A | Relaxed |
| pCDF | ~20-40 | CloDF13 (CDF) | D | Relaxed |
This table defines common cloning vectors, their copy number, ori, and incompatibility groups. Note the A -C compatibility grouping is an arbitrary designation, and plasmids from the same incompatibility group should not be co-transformed.
+Actual copy number varies. See below for additional considerations.
*Requires pir gene for replication (reference).
**F1 is a phage-derived ori that allows for the replication and packaging of ssDNA into phage particles. Plasmids with phage-derived ori's are referred to as phagemids.
Other factors that affect plasmid copy number
Although the sequence and regulation of the ori dramatically affect the copy number of a plasmid, other external factors contribute as well. These considerations are especially useful to keep in mind if you are planning to purify your plasmid DNA:
The insert
- Bacteria tend to maintain fewer copies of plasmids if they contain large inserts or genes that create a toxic product.
The E. coli strain
- Most E. coli strains can be used to propagate plasmids, but endA- E. coli are best for high yields of plasmids.
Growth conditions
- The amount of aeration, temperature, culture volume, antibiotic, and medium can all affect copy number. Some ori's are temperature sensitive; others ori's can be "tricked" into amplifying more copies with the addition of chloramphenicol – make sure your growth conditions aren't working against you!
The culture inoculum
- Freshly streaked bacteria have higher copy numbers – for optimal results always pick a single colony and do not subculture directly from glycerol stocks, agar stabs, or liquid cultures.
- Freshly transformed cells also give a higher plasmid yield that colonies streaked from a glycerol stock.
- Incubation for 12-16 hours tends to give higher copy numbers since the bacteria have just reached stationary phase, but the cells have not started to die off.
Have any questions? Need something clarified? If so please, post in the comments.
References
Chatterjee A, Sun SB, Furman JL, Xiao H, Schultz PG. A versatile platform for single- and multiple-unnatural amino acid mutagenesis in Escherichia coli. Biochemistry. 2013 Mar 12;52(10):1828-37. doi: 10.1021/bi4000244. Epub 2013 Feb 27. PMID: 23379331; PMCID: PMC3855549.
Topics: Plasmid Elements, Plasmids 101, Plasmids






Leave a Comment