For safety and production reasons, multiple plasmids with many components are used to package lab grown viruses. It can be intimidating at first to look at so many plasmid maps and protocols! In reality, there are many shared components across viruses (AAV, lentivirus, etc.) and their functions are more intuitive than you might think. So, if you’ve ever wondered what goes into ‘making virus’ or what gag and pol stand for, we demystify all components of viral vectors here!
Need Virus? Check out Addgene's Viral Service!
Types of viral vectors
Of the many viruses out there, only a small subset are commonly used as tools in the lab. We will discuss the gamma-retrovirus, lentivirus, adenovirus, and adeno-associated virus (AAV). The components of these vectors are also split across several plasmids (often three), for safety purposes. The primary plasmid with the delivered cargo (often a gene) is called the transfer plasmid while the other plasmids with packaging or replication components are called helper plasmids. Below, we’ll breakdown the vector components first by virus and then by function.
Lentivirus/gamma-retrovirus
Lentiviruses are a class of retrovirus (so all lentiviruses are retroviruses, but not all retroviruses are lentiviruses.) This means that lentivirus and gamma-retrovirus share a lot of core components, so we will discuss them together.
Gene expression
The reason for most viral experiments is to deliver a genetic cargo, typically an expression construct, to cells/organisms. This is often cDNA, shRNA, or something similar, and is contained on the transfer plasmid. If expression is the goal, a promoter should flank this component as well. On both sides of this promoter-gene combo are two long terminal repeats (LTRs). These LTRs define the limit of what will be packaged. The 5’ LTR and 3’ LTR, respectively, act as RNA polymerase II promoter and terminator regions and are slightly different in sequence. Both LTRs do share common repeat regions in addition to their unique regions as well, though.
Viral packaging and entry
The genetic cargo requires viral packaging to infiltrate and integrate into the host genome. First, it needs to be virally enveloped which is achieved by expression of the vesicular somatitis virus G glycoprotein (VSVG). VSVG is a broad tropism envelope protein that can also be pseudotyped to alter infectivity. Next, the virus must assemble and have appropriate factors displayed on the outside of the particle to interact with the host. This is the role of Gag proteins: they contain the matrix, capsid, and nucleocapsid viral components needed for viral packaging, assembly, and display.
Pol, the last component, contains the reverse transcriptase and integrase components to convert the cargo from RNA to DNA and permanently insert the cargo into the host genome.
Lentiviral-specific components
Lentiviruses have gone through their fair share of lab evolution. There are 1st, 2nd, and 3rd generation plasmids for their production. As such, there are bonus components to these vectors that aren’t reflected in the common components outlined above for gamma-retroviruses. While not required for lentiviral production, they can be helpful for certain applications and aid in ease of use. Our lentiviral guide outlines these components.
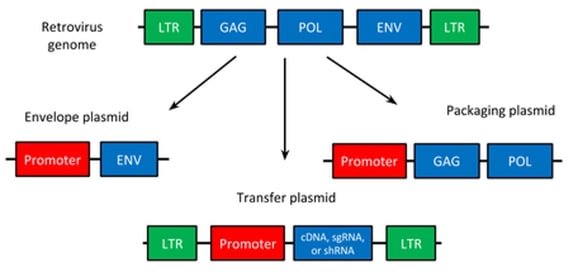
|
| Fig. 1 - Viral vector components of retrovirus |
Adeno-associated virus
AAV’s elements are quite different from gamma-retrovirus, but they perform very similar functions to infiltrate the host cell and deposit viral cargo.
Gene expression
While AAV often does not integrate into the host genome (more on this below), it’s still used to deliver gene expression constructs. Sandwiched between two inverted terminal repeats (ITRs) is the promoter and cargo, in that order. This will all be ssDNA within the viral particle. The ITRs base pair with each other to allow DNA synthesis of the complementary strand to yield dsDNA. Unlike LTRs, ITRs are perfectly symmetrical to allow for this pairing to occur. Like LTRs, the ITRs define the limit of what will be delivered and expressed in the host cell from the transfer plasmid.
Viral packaging and entry
The viral packaging proteins for AAV are rep and cap. Rep is required for viral replication and also aids in infrequent genome integration at the AAVS1 locus in humans. Cap is a structural capsid protein that helps with virion assembly and particle release from late endosomes when AAV is trafficked. Rep and cap mediate AAV pseudotyping which allows for altered tropism of AAV to target different tissue or cell types. AAV also requires some back up from adenovirus genes – E4, E2a, and VA – which are supplied on a helper plasmid and mediate efficient viral replication. More information on these viral genes is available in the adenovirus section below!
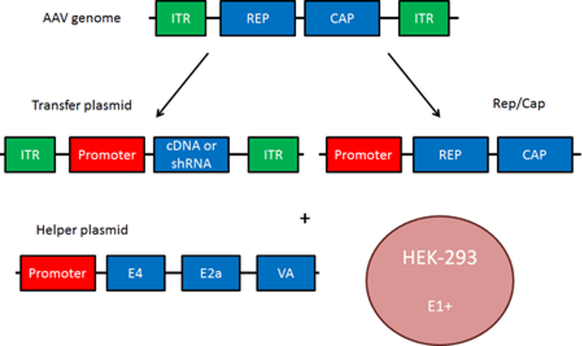 |
| Fig. 2 - Viral vector components of AAV |
Adenovirus
Adenoviruses have quite a bit in common with AAV, just like lenti and gamma-retroviruses have a lot in common with each other. Below we will outline the common features as well as the differences within their viral elements.
Gene expression
Adenovirus is a linear, dsDNA virus naturally. It has ITRs, just like AAV, which encompass the expression construct and promoter that you wish to deliver to your target cells. In adenovirus, the ITRs serve as a type of origin of replication to initiate replication of the dsDNA virus.
Viral packaging and entry
Adenovirus is quite large and has more helper plasmid components than the other viruses discussed. LXs/Iva2 are primarily capsid assembly proteins required for packaging of the virus into particles. E2A/E2B are responsible for viral replication, with E2B being the primary viral polymerase. E4/E1A activate transcription of other relevant viral genes (including the ones listed previously). E3/E1B suppresses both apoptosis and the host immune response to the virus to allow it to evade detection by the host. Of note, recombinant adenovirus is missing the endogenous E1 gene, making it replication deficient. This is typically supplied by the production cell line (HEK-293) during production.
Fun fact: There are 57 human adenovirus types, but most vectors are based on adenovirus 5 (Ad5).
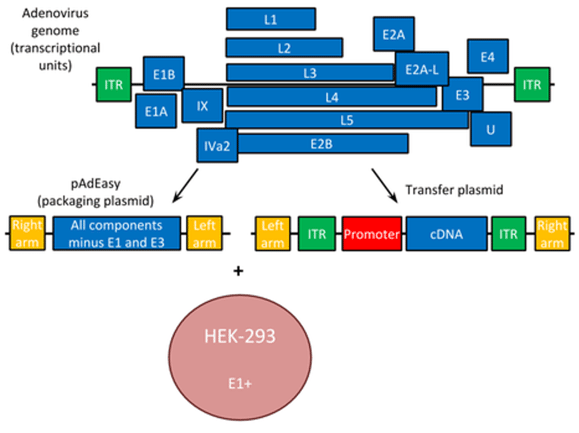
|
| Fig. 3 - Viral vector components of adenovirus |
Building viral vectors
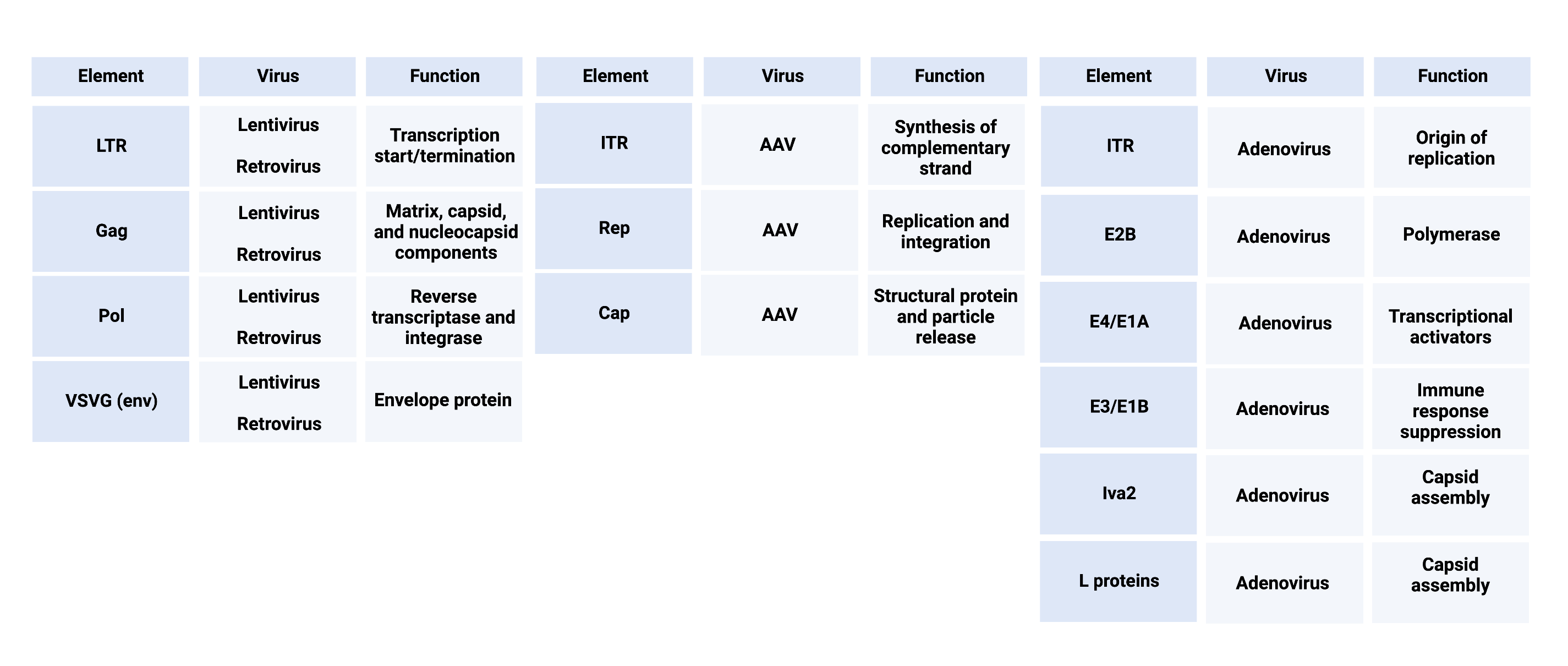
Addgene has many viral vectors and helper plasmids for all the viruses discussed above (gamma-retrovirus, lentivirus, adenovirus, and adeno-associated virus (AAV). We also offer viral prep services for some of the plasmids in our collection. Want to make a vector construct on your own? No problem, we are here to help with resources from AAV variables that matter to adenovirus production tips. For quick review, all of the elements discussed are compiled for reference below. Happy virus making!
References and Resources
Kulanayake, S., Tikoo, S. K. Adenovirus Core Proteins: Structure and Function. Viruses. 2021 13(3):388. DOI:10.3390/v13030388
Coffin J.M., Hughes S.H., Varmus H.E. Retroviruses. Cold Spring Harbor (NY): Cold Spring Harbor Laboratory Press; 1997. Principles of Retroviral Vector Design. Available from: https://www.ncbi.nlm.nih.gov/books/NBK19423/
Resources on Addgene.org
- Addgene's most popular lentiviral plasmids
- Addgene's most popular retroviral plasmids
- Addgene's collection of AAV plasmids
- Addgene’s collection of adenovirus plasmids
Resources on the Addgene blog
Topics: Viral Vectors 101





Leave a Comment