Western blots. ELISAs. Immunofluorescence. What do all of these techniques have in common? They all typically require secondary antibodies, frequently of the mouse or rabbit variety. While antibodies certainly aren’t “broken,” their production does require continued animal sacrifice. Could there be an alternative method for immunodetection? Enter the Görlich lab and their anti-mouse and -rabbit IgG secondary nanobodies toolbox. Nanobodies are like tiny antibodies which work just as well, if not better, than antibodies for all of the above listed molecular techniques, but they can also be expressed in bacteria and extracted with common protein purification methods. Read on to learn more about nanobodies and how their structure and function compare to IgG antibodies, as well as how to produce them for use in your lab.
- Structural Comparison
- Functional Comparison
- Advantages of Nanobodies
- How to Use the Nanobody Toolbox
- Find Nanobody Plasmids
Structural comparison
As the name suggests, nanobodies are smaller antibodies.They are derived from an unusual type of IgG antibody called a heavy chain antibody (HCab), which are unique to camels, llamas, alpacas and other camelids. In terms of structure, HCabs are like a pared down version of a standard IgG. See below for a comparison of nanobody, HCab, and traditional IgG antibody structures.
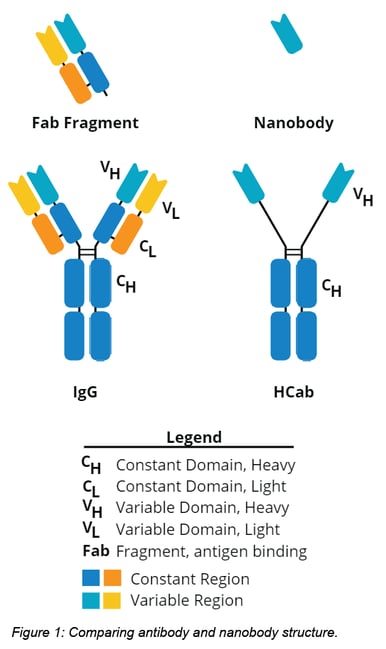
Functional comparison
Strong but Large: Advantages of Antibodies
One of the main reasons antibodies are great indirect immunodetection tools is their strong signal strength. This signal strength is due in large part to their larger size (~10-15 nm), which allows for the following to occur:
A) Multiple polyclonal secondary IgGs can bind to different epitopes of a primary antibody.
B) Each arm of the antibody has an antigen binding site for a total of 2 binding sites per antibody.
C) IgG’s are big enough that multiple labels (green circles in figure 2 below) can be attached to each antibody.
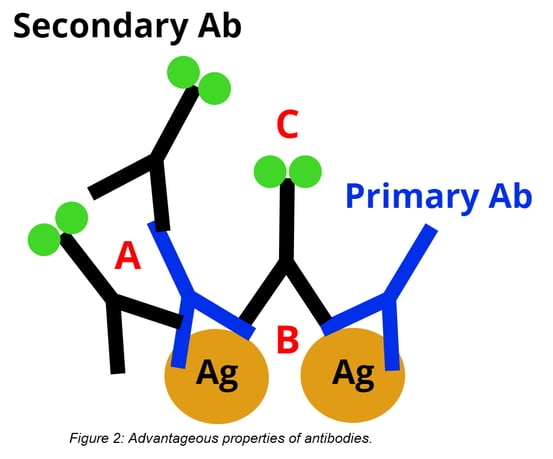
But being big is a double-edged sword. An antibody’s large size creates more distance between the target antigen and the fluorescent label, which can reduce the potential image resolution of super-resolution fluorescence microscopy. Their size also limits their ability to penetrate tissue. Because antibodies are large and bivalent, primary and secondary antibodies cannot be incubated together without forming large oligomeric complexes. Antibodies are also too large to be expressed in bacteria, which means their production requires animal sacrifice and precludes genetic engineering of a tag or fusion with a reporter enzyme.
For multicolor staining, careful consideration must be used when selecting which antibodies to use together. It’s best to select primary antibodies raised in different species, or at least from different IgG subclasses, so that secondary antibodies exclusively recognize only one primary antibody. This process can be avoided direct antibody fluorophore conjugates are used, but these are usually more expensive than unconjugated primary antibodies.
| Reagent | Pros | Cons |
| Antibodies |
|
|
| Nanobodies |
|
|
Small but Mighty: Advantages of Nanobodies
Although antibodies have been the titans of immunodetection, nanobodies may be better suited for the job. As single domain proteins, they can be expressed in bacteria, making nanobodies a recombinant and renewable anti-IgG reagent. Their small size also allows for better tissue penetration and decreases the distance between a fluorescent tag and the target antigen to about 2 nm, which can lead to higher resolution for super-resolution microscopy. Nanobodies can be labeled with up to 3 dyes, yielding similar signal strength to a standard IgG secondary antibody (Pleiner et al figures 4A-C). And since they have limited cross-reactivity between different IgG subclasses and different species, nanobodies are useful for colocalization labeling of up to 3 targets (Pleiner et al figure 4D).
Unlike large secondary antibodies, small nanobodies do not oligomerize with primary antibodies when incubated together. At the bench, this results in a shorter staining protocol because the primary antibody can be “labeled” by incubating it with a tagged nanobody. After this pre-incubation step, the primary antibody-nanobody mix can be added to your sample, thus eliminating the secondary antibody incubation. See figure 4A below for comparison of this one-step staining with nanobodies vs two-step staining with antibodies.
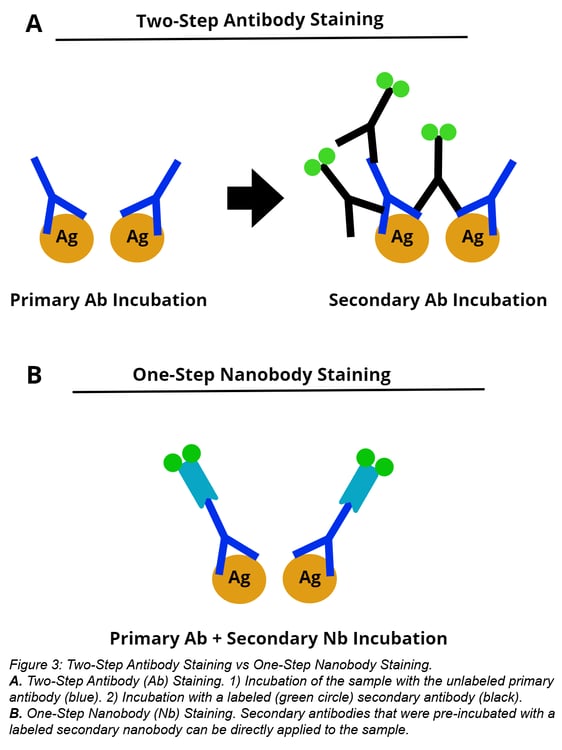
Nanobodies also simplify multicolor staining experiments because they don’t require using primary antibodies from different IgG subclasses. Instead, you can pre-incubate individual primary antibodies with secondary nanobodies conjugated with the desired fluorophores. In figure 4B below, triple colocalization is possible even when staining cells with three IgG1 antibodies. Antibodies were individually incubated with a secondary nanobody conjugated with Alexa Fluor 488, Alexa Fluor 568, or Alexa Fluor 647 before application to the sample. Staining with this multi-color staining workflow yields similar localization patterns as cells stained for one target (compare nanobodies staining in figure 4A vs 4B).
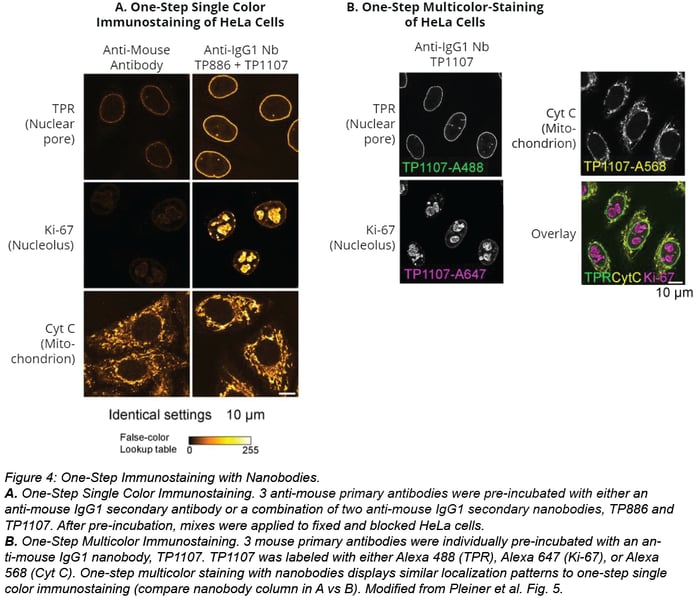
How to use the secondary nanobody toolbox
Are you ready to start using nanobodies for your own research? Great! Pleiner et al. have done the hard work for you by immunizing an alpaca, isolating HCab antibodies from its serum, and meticulously screening, optimizing, and characterizing several anti-mouse and -rabbit nanobodies. If you’re interested, you can learn more about how nanobodies are identified in this review. If you just want to use the nanobodies described in Pleiner et al, the key steps for generating nanobodies are outlined below; no alpaca required.
Decide Which Nanobody To Use:
The table below summarizes currently available nanobodies from Pleiner et al. Before picking a secondary nanobody, it’s important to review a few characteristics of your primary antibody: species it was raised in and its IgG subclass. Once you know the answer to these two questions, refer to this table to find the right nanobody for your work.
| Species | Antibody Subclass | Nanobody | Addgene Plasmid |
| Rabbit | N/A* | TP897 | |
| Mouse | IgG1 | TP886 | pTP943 (3x cysteines) |
| Mouse | IgG1 | TP1107 | |
| Mouse | IgG2a | TP1129 | pTP1005 (3x cysteines) |
| Mouse | Kappa | TP1170 |
*Rabbits have only one IgG subclass.
Express the Nanobody In Bacteria:
Table 2 also has a link to the plasmid you’ll need to express your nanobody of choice in bacteria. Notice that nanobodies come in 1x and/or a 3x cysteine varieties. This refers to the number of cysteine sites the nanobody has available for conjugation to a specific label (see step 4 for more information).
Purify the Nanobody With A Nickel Column:
Lyse the bacteria expressing your nanobody and run the lysate over a nickel column. Nanobodies will bind to the column due to their N-terminal His14-bdNEDD8 tag. After several washes, this tag can be cleaved by the bdNEDP1 protease, thus releasing purified nanobodies from the column. bdNEDP1 protease can be generated using plasmid pDG02583.
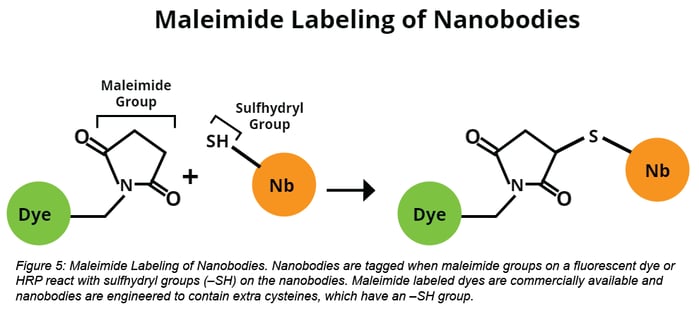
Label the Nanobodies:
Purified nanobodies are then tagged with a fluorescent dye or HRP via maleimide-labeling. Maleimide-labeling occurs when a maleimide group forms a covalent bond with a sulfhydryl group (–SH), such as that found in cysteine. Maleimide-conjugated HRP can be found here and several maleimide-conjugated dyes are also commercially available.
References
1. Pleiner, T., Bates, M., & Görlich, D. (2017). A toolbox of anti–mouse and anti–rabbit IgG secondary nanobodies. The Journal of Cell Biology. PubMed PMID: 29263082
- Find plasmids from this publication at Addgene.
2. Bates, M., Böhning, M.I., Chug, H., Görlich, D., Lee, C., Pleiner, T., Schliep, J.E., Stark, H., Trakhanov, S., & Urlaub, H. (2015). Nanobodies: site-specific labeling for super-resolution imaging, rapid epitope-mapping and native protein complex isolation. eLife. PubMed PMID: 26633879 PubMed Central PMCID: PMC4755751
3. Muyldermans, Serge. “Nanobodies: natural single-domain antibodies.” Annual review of biochemistry 82 (2013): 775-97. PubMed PMID: 23495938.
Additional Resources on the Addgene Blog
- DIY DNA ladders from Penn State University
- Use Nanobodies to Control Protein Activity
- Learn more about protein tags
- Read about E. coli strains for protein expression
Additional Resources on Addgene.org
Additional Resources
Looking for monoclonal primary antibodies to use with the secondary nanobody toolbox? Check out the NIH’s Developmental Studies Hybridoma Bank (DSHB). 1 ml of antibody-containing hybridoma supernatant is only $40, significantly cheaper than commercially available antibodies.
Topics: Plasmids 101, Plasmids, Antibodies






Leave a Comment