In a lab, you may have heard the phrase gram negative or positive being used to describe a species of bacteria, but what does it actually mean? What relevance does it have on the structure of a bacteria species and how it can be used in a lab? Here we’ll be talking about the ins and outs of this classification system, including its history, biology, and usages in the lab.
History
In 1884, while studying infected lung tissue, a Danish biologist named Christian Gram realized that certain stains were retained only in some species of bacteria. The stain colored one type, Streptococcus pneumoniae, while the alcohol used in the staining process bleached another, Klebsiella pneumoniae. After realizing this was a repeatable phenomenon, Gram began using it as a classification system. Eventually, any bacteria that retained the stain became known as gram positive, and the decolorized as gram negative. A German scientist, Carl Weigert, later expanded on Gram’s discovery, adding a second stain, safranin, as a counterstain to help visualize the bleached bacteria. The chemistry behind Gram’s crystal violet stain and the ion interactions that cause the stain retention in gram positive bacteria was discovered in 1983. The differences between gram positive and negative bacteria is due to differences in cell wall composition.
Cell wall structure
Gram positive bacteria, or monoderms, have a single thick peptidoglycan membrane. Common gram positive bacteria include staphylococci and streptococci. Gram negative bacteria, or diderms, have a thinner peptidoglycan membrane and an outer lipid membrane. Common gram negative bacteria include salmonella and E. coli. Because of their thicker peptidoglycan layer, gram positive bacteria trap the crystal violet stain within their membrane, while the stain is washed from the gram negative bacteria’s thinner membrane.
Procedure
The stain is a pretty simple process. First, flood the heat-fixed smear with the crystal violet stain for 1 minute. Rinse the slide briefly with tap water. Flood the slide with Gram's iodine solution. Rinse briefly with tap water. Add ethyl alcohol or acetone drop by drop onto the smear until the alcohol runs clear and immediately rinse. Flood the smear with the safranin counterstain for 30 - 60 seconds. Rinse the slide in tap water until the runoff is clear, blot gently to dry, and then view under an oil immersion using a light microscope. Gram positive bacteria will be stained blue or purple, and gram negative bacteria will be stained red or pink, as shown in the image below.
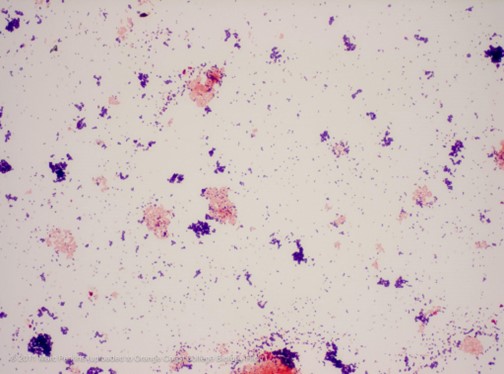 |
| An example of Gram positive (purple) and Gram negative (red/pink) staining. Image credit: Mark Perkins https://www.flickr.com/photos/occbio/7092385559 |
For more specific procedural instructions, including timing, preparing the smear, and mixing the stains and solutions used, check out these resources.
- Bruckner, M. Gram Staining. https://serc.carleton.edu/microbelife/research_methods/microscopy/gramstain.html
- Smith, A. & Hussey, M. Gram Stain Protocols. American Society for Microbiology (2005).
- https://asm.org/getattachment/5c95a063-326b-4b2f-98ce-001de9a5ece3/gram-stain-protocol-2886.pdf
- Steward, K. Gram Positive vs Gram Negative. Technology Networks (2019) https://www.technologynetworks.com/immunology/articles/gram-positive-vs-gram-negative-323007
How the stain works
Crystal violet (CV) is water soluble and can enter the peptidoglycan layer of both bacteria types. In water, the dye dissociates into CV+ and Chloride (Cl-) ions. When the negatively charged iodine is added, it interacts with the CV+, creating a complex within the peptidoglycan layer, staining the cell purple. When the alcohol is added, it dehydrates the peptidoglycan layer, trapping the large dye complex created by the interaction of CV+ and I- in the thick multilayered membrane of the gram positive bacteria. In a gram negative bacteria, the alcohol destroys the outer lipid membrane, and the thin inner peptidoglycan membrane is unable to retain the stain, leading to its decolorization.
In contrast, counterstain, safranin, is a light and water soluble dye that is able to enter the peptidoglycan layer. It is not dark enough to affect the gram positive bacteria, which is already stained by the crystal violet dye, but is easily retained in the decolorized gram negative bacteria, staining them red or pink. At the end of the staining procedure, gram positive bacteria are stained purple or blue, and gram negative bacteria are stained red or pink.
Purpose
Gram staining can be used in medical settings to determine the general type of bacteria present in an infection. This information can then be used to determine effective courses of treatment, since gram negative and positive bacteria are permeable to different classes of antibiotics. The classification system is also useful in a laboratory setting. When designing a plasmid to be inserted into a specific bacterium, it can be important to consider the type of bacteria it is being used in, and how components of the plasmid should change based on bacteria type. The gram positive and negative classification system is incredibly helpful in visualizing differentiations between bacteria, leading to safer medicine and more successful experiments. It’s something that all scientists should understand when stepping into a lab.
Anna Kamens is a science communication summer intern at Addgene and a rising third-year at Old Dominion University, who is majoring in marine biology and minoring in studio arts photography.
References and resources
References
Silhavy, TJ., Kahne, D. & Walker S. The Bacterial Cell Envelope. Cold Spring Harb Perspect Biol. 2(5) a000414 (2010).
Cleveland Clinic. Gram Stain. https://my.clevelandclinic.org/health/diagnostics/22612-gram-stain
Hardy, J. Gram’s Serendipitous Stain. https://hardydiagnostics.com/wp-content/uploads/2016/05/Hans-Christian-Gram.pdf
Smith, A. & Hussey, M. Gram Stain Protocols. American Society for Microbiology (2005).
https://asm.org/getattachment/5c95a063-326b-4b2f-98ce-001de9a5ece3/gram-stain-protocol-2886.pdf
Steward, K. Gram Positive vs Gram Negative. Technology Networks (2019) https://www.technologynetworks.com/immunology/articles/gram-positive-vs-gram-negative-323007
More resources on the Addgene blog
Plasmids 101: What is a plasmid?
Plasmids 101: Blue-white screening
Plasmids 101: Common lab E. Coli strains
Topics: Plasmids 101, Molecular Biology Protocols and Tips, Plasmids





Leave a Comment