This post was contributed by Adam Chin-Fatt, a Ph.D. student at the University of Western Ontario. Adam summarizes Zalatan JG, et al.'s recent paper, "Engineering Complex Synthetic Transcriptional Programs with CRISPR RNA Scaffolds." Adam has also created a video to help scientists visualize the concepts discussed in the paper.
 The transcriptional control of multiple loci is deftly coordinated by the eukaryotic cell for the execution of many complex cellular behaviors, such as differentiation or metabolism. Our attempts to manipulate these cellular behaviors often fall short with the generation of various flux imbalances. The conventional approach has typically been to either systematically delete/overexpress endogenous genes or to introduce heterologous genes, but the trend of research has shifted in recent years toward tinkering with regulatory networks and multiplex gene control. However, these approaches are often met with the challenges of regulatory bottlenecks and their scope is limited by the lack of well characterized inducible promoters. Far removed from the bio-industry’s vision of ‘biofactories’, most successes in metabolic engineering have been limited to the overexpression of various metabolites in Escherichia coli or Saccharomyces cerevisiae with few techniques that are easily transferrable across host species or metabolic pathways. A new study takes us one step closer to the vision of metabolic biofactories by demonstrating the use of CRISPR-based RNA scaffolds to mimic natural transcriptional programs on multiple genes.
The transcriptional control of multiple loci is deftly coordinated by the eukaryotic cell for the execution of many complex cellular behaviors, such as differentiation or metabolism. Our attempts to manipulate these cellular behaviors often fall short with the generation of various flux imbalances. The conventional approach has typically been to either systematically delete/overexpress endogenous genes or to introduce heterologous genes, but the trend of research has shifted in recent years toward tinkering with regulatory networks and multiplex gene control. However, these approaches are often met with the challenges of regulatory bottlenecks and their scope is limited by the lack of well characterized inducible promoters. Far removed from the bio-industry’s vision of ‘biofactories’, most successes in metabolic engineering have been limited to the overexpression of various metabolites in Escherichia coli or Saccharomyces cerevisiae with few techniques that are easily transferrable across host species or metabolic pathways. A new study takes us one step closer to the vision of metabolic biofactories by demonstrating the use of CRISPR-based RNA scaffolds to mimic natural transcriptional programs on multiple genes.
Modifying the CRISPRi system for multidirectional control
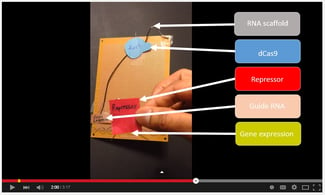
The CRISPR interference (CRISPRi) model consists of a catalytically dead Cas9 protein (dCas9) that lacks endonuclease activity coupled with a customizable single guide RNA (sgRNA) that bears complementarity to a 20bp target sequence (Qi et al. 2013). Modifying the CRISPRi system for more flexible multidirectional control, a new study repurposes the sgRNA to include modular synthetic RNA scaffolds that can recruit various transcriptional regulators (Zalatan et al. 2015). These scaffolds are stretches of non-coding RNA interspaced with linkers that can fold into a secondary structure that can bind specific proteins via aptamer domains. Combined with dCas9, this new scaffold RNA (scRNA) will bind to a PAM-adjacent target nucleotide sequence then subsequently recruit a transcriptional regulator to modulate expression at that locus. The use of regulators allows a greater degree of control of desired expression levels dependent upon the type of transcriptional regulator or the number of designed scaffold binding sites. With hundreds of characterized aptamer domains available, an unprecedented level of flexible tunability of transcription is possible.
From a design perspective, this is appealing since the RNA scaffold modules are comparatively small allowing multiple sgRNA constructs to be introduced simultaneously in a single plasmid. The functionality of the RNA scaffolds follows the rules of Watson-Crick base pairing, allowing for rational, straightforward design. Since the technique relies on base pairing, it has promise to be host-independent, beyond the oft published metabolic engineering realm of S. cerevisiae and E. coli.
The utility of simultaneous and multidirectional multiplex control via scRNA design is readily apparent in metabolic engineering. The authors elegantly demonstrated the potential of this to control a metabolic network in S. cerevisiae by using three RNA programs to differentially alter the levels of five genes that encode biosynthetic enzymes for violacein, a distinctive pigmented protein. By altering the transcriptional programs, the authors sequentially rerouted the metabolic flux to generate differently pigmented derivatives of violacein.
Using dCas9 as an on/off switch
The multiple scRNAs all individually utilize the same dCas9 protein. By using an inducible promoter to modulate dCas9 expression, multiple scRNA transcriptional programs can be regulated with dCas9 as a master ON/OFF switch. This will likely prove particularly useful in avoiding toxic intermediate buildup as well as controlling cell fate decisions. By using an orthogonal dCas9 protein that will still recognize guide RNA but with a different dCas9 binding module, alternative sets of transcriptional programs can be layered with distinct programs controlled by each dCas9 regulatory switch. By no means limited to the field of metabolic engineering, this is such a powerful and flexible technique that it should be on the radar of every molecular microbiologist.
Summary
- The single guide RNA in the CRISPRi system can be repurposed to include modular RNA scaffolds.
- These RNA scaffolds can recruit various transcriptional regulators to alter gene expression at the targeted site.
- Multiple scaffold RNAs can be designed to allow simultaneous, multidirectional and tunable control of gene expression.
- This strategy can be applied to effectively change metabolic flux.
- The dCas9 protein and orthogonal variants can be used as regulatory ON/OFF master switches to induce multiple transcriptional programs.
Thank you to our guest blogger!
 Adam Chin-Fatt is a Ph.D. student at the University of Western Ontario studying plant-based recombinant pharmaceuticals. Follow him on Twitter @AdamChinFatt
Adam Chin-Fatt is a Ph.D. student at the University of Western Ontario studying plant-based recombinant pharmaceuticals. Follow him on Twitter @AdamChinFatt
References
- Qi, L. S. et al. Repurposing CRISPR as an RNA-guided platform for sequence-specific control of gene expression. Cell, 152(5), 1173-1183. (2013). PubMed.
- Zalatan, J. G. Engineering Complex Synthetic Transcriptional Programs with CRISPR RNA Scaffolds. Cell, 160(1), 339-350 (2015). PubMed.
Find the Plasmids @Addgene:
- Plasmids for CRISPRi are described here: http://www.addgene.org/crispr/qi/
- Plasmids described in Zalatan et al. Cell (2015) can be found here: https://www.addgene.org/browse/article/9593/
- Plasmids described in Qi et al. Cell (2013) can be found here: https://www.addgene.org/browse/article/6441/
Resources:
- An open source library of characterized aptamers and supporting publication: http://www.freebase.com/base/aptamer
- Cruz-Toledo, J. et al. Aptamer base: a collaborative knowledge base to describe aptamers and SELEX experiments. Database, bas006 (2012). PubMed.
- For a stepwise protocol on designing and using RNA scaffolds: Delebecque, C. J. et al. Designing and using RNA scaffolds to assemble proteins in vivo. Nature protocols. 7(10) 1797-1807 (2012). PubMed.
Topics: CRISPR, Other CRISPR Tools







Leave a Comment