Optogenetics gives you the power to control cells or organisms with the flip of a switch. You may be familiar with popular light-sensitive ion channels used to control activities like neuronal signaling — think of a mouse with an LED brain implant or a worm wiggling back and forth under blue light — but there’s a lot more to optogenetics than neuronal activity. Researchers have designed a variety of photoswitchable elements into many other types of proteins. Here, we’ll highlight a few recently-deposited optogenetic tools to control the cytoskeleton and cell membrane dynamics.
First, a quick refresher.
The tools below use the following optogenetic modules:
- VVDfast homodimerizes under blue light
- iLID-SspB heterodimerizes under blue light
- BcLOV4 undergoes a conformational change and binds the plasma membrane under blue light
- CRY2-CIB1 heterodimerizes or forms clusters under blue light
Check out our Optogenetics Guide to learn more about how these modules work, and read on to learn how they are put to use. And now, onto our optogenetic tools!
Opto-katanin: a light-powered microtubule-cutting sword
Microtubules are one of the most important components of the cytoskeleton. They provide cells with physical structure, separate chromosomes during mitosis, support transport of organelles and vesicles, and more. Although several strategies are available to disrupt microtubules in cells, they have various disadvantages and limitations – for example, microtubule-destabilizing drugs are impossible to target to specific subcellular regions, laser ablation can damage other cellular structures, and genetic mutations are not easily reversible. Late last year, the Akhmanova Lab published a new optogenetic tool for localized microtubule disassembly, Opto-katanin (Meiring et al., 2022), that overcomes many of those limitations (Figure 1).
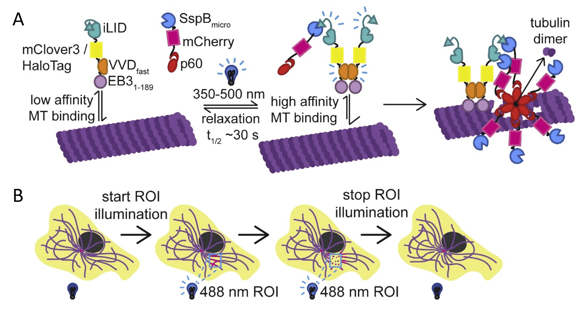
Figure 1: Opto-katanin induces fast, localized microtubule severing. A) Schematic of Opto-katanin constructs and activity. EB3N-VVDfast-iLID (anchor) and SspB-mCherry-p60 (katanin) are recruited to microtubules under blue light, removing tubulin dimers from the microtubule body. B) Schematic of Opto-katanin’s use in cells. Images adapted from Meiring et al., 2022, under CC-BY 4.0 license.
A naturally occurring microtubule-severing protein, Katanin is of course named for the samurai sword called a katana. Opto-katanin consists of two separate blue light-sensitive systems, VVDfast to control recruitment of an “anchor” to microtubules and iLID-SspB to control the recruitment of katanin to that anchor. So, you’ll need to express both the EB3N-VVDfast-iLID anchor module and the SspB-mCherry-p60 katanin module, which are also available as lentiviral plasmids if you want to make a stable cell line. This design allows highly localized, rapid and reversible microtubule severing and has very low dark-state activity and toxicity. All of that means this light-powered sword truly is an elegant tool.
The authors demonstrated its use to probe cell mechanics, vesicle transport, and cell division, but given the long list of roles played by microtubules, Opto-katanin will surely be useful for a wide variety of experiments.
Opto-RhoGEFs manipulate cell morphology and adhesion
But what if you’re interested in that other most-important component of the cytoskeleton – actin? Actin dynamics control cell morphology, movement, and more, often driven by Rho family signaling pathways. Rho guanine-nucleotide exchange factors (GEFs) activate their partner Rho GTPase, leading to behaviors like lamellipodia or filopodia formation, cell contraction, or other morphological changes, depending on the specific Rho GTPase/GEF pair (Ridley, 2016). Taking control of a RhoGEF is a great way to stimulate and study these physical processes.
Joachim Goedhart and colleagues in the Gadella Lab devised a set of optogenetic switches for the GEFs ITSN1, TIAM1, or p63RhoGEF (which activate the Rho GTPases Cdc42, Rac1, and RhoA, respectively) using the iLID system (Mahlandt et al., 2023). These Opto-RhoGEFs provide a reversible and non-invasive way to activate a RhoGTPase by recruiting its GEF to a specific area at the plasma membrane (Figure 2). Just express the plasma membrane-localized iLID along with your chosen SspB-tag-GEF and your cells will be ready for the spotlight.
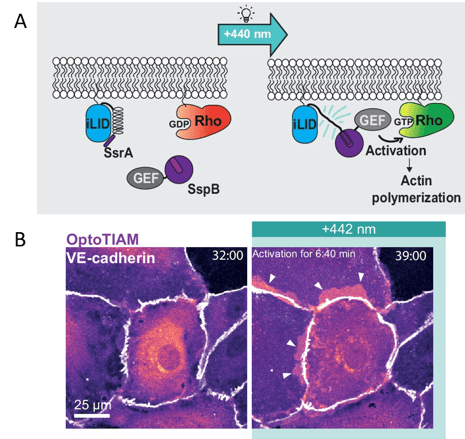
Figure 2: Opto-RhoGEFs to control Rho GTPase activity. A) Schematic of Opto-RhoGEF design and activity. B) OptoTIAM (magenta) expressed in a monolayer of endothelial cells, showing cell protrusions and overlaps (arrows) after blue light illumination. Scale bar 25 µm. Images adapted from Mahlandt et al., 2023, under CC-BY 4.0 license.
Mahlandt et al. used these Opto-RhoGEFs to study cell junction integrity in the vascular barrier of the endothelium, but they could be applied to study other actin-dependent processes in cells and tissues. And, this Opto-RhoGEF design could be extended to other GTPases.
The Chow Lab has also generated a variety of membrane-recruitment optogenetic tools for manipulating Rho GTPases based on BcLOV4. These single-component systems take advantage of BcLOV4’s light-induced binding of the plasma membrane. In previous publications, they described tools to control formation of lamellipodia and contractile stress fibers, and recently developed a full toolbox and workflow for generating new Rho GTPase optogenetic systems (Berlew et al., 2022).
Stopping traffic with CIB1-Rabs
Instead of using an optogenetic module to activate or recruit your protein of interest, maybe you want to inhibit it. Restricting the effect to a specific time or location can be critical when, say, the gene is essential during development and a knockout is not feasible or when the protein of interest supports a variety of processes in different cell types. One great way to do this is to use CRY2-CIB1 clustering. Fuse either CRY2 or CIB1 to your protein of interest and express the other separately, then use blue light to generate CRY2-CIB1 oligomers that trap the fusion protein away from its usual location, preventing it from carrying out its proper function (Figure 3).
The Hehnly Lab used such a strategy to dissect the roles of three Rab proteins during a key stage of zebrafish development (Aljiboury et al., 2023). Rab family proteins support vesicle trafficking and organelle dynamics, but with dozens of family members and many functions it can be difficult to unravel their specific roles. Zebrafish are an excellent model for optogenetic studies since they are transparent and the developing embryo is accessible for injection of plasmids and other materials.
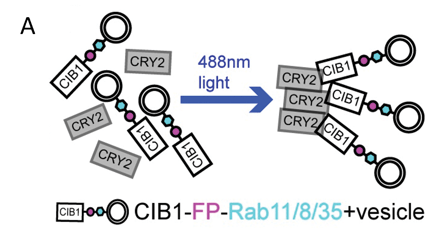
Figure 3: Optogenetic inhibition of Rab protein vesicle trafficking via CRY2-CIB1 clustering. Image adapted from Aljiboury et al., 2023, under CC-BY 4.0 license.
Building off their earlier work on Rab11, the team used optogenetic clustering of Rab8, Rab11, and Rab35 to understand their roles in forming the cilia that help establish left-right organization of the embryo (Aljiboury et al., 2023). It’s a great example of how optogenetics allows you to disrupt and study a protein’s function at a very specific place and time. Future studies with these CIB1-Rabs in other contexts are sure to reveal even more insights.
Super-duper lipid editor
And if recruiting proteins to the cell membrane isn’t powerful enough, what about altering the chemical composition of the membrane itself? The Baskin Lab previously published OptoPLD, an optogenetically controlled phospholipase D (PLD). Now they present a set of variants called SuperPLDs with 10, 30, 50, and 100 times the activity of the original, generated using directed evolution (Tei et al., 2023).
PLD can hydrolyze phosphatidylcholine (PC), the predominant membrane lipid, to phosphatidic acid (PA) or it can swap phosphatidylchonine’s choline head group for other natural or synthetic head groups. The new SuperPLDs work with high efficiency in mammalian cells and in vitro, opening up a wide realm of possibilities. The CRY2-SuperPLD module is optogenetically recruited by different CIB constructs to the plasma membrane or lysosomes (the earlier article also provided CIB constructs for targeting Golgi, ER, and endosome membranes) (Figure 4). The authors used SuperPLD to manipulate phosphatidic acid signaling (such as Hippo, AMPK and mTOR pathways) and to introduce unnatural head groups for applications like fluorescent labeling via click chemistry.
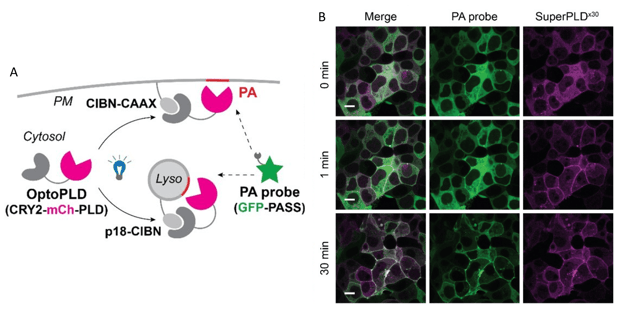
Figure 4: SuperPLD hydrolyzes membrane PC to PA. A) OptoPLD is recruited to plasma membrane or lysosomes and its activity can be monitored by phosphatidic acid probe GFP-PASS. B) Time series of HEK 293T cells expressing plasma membrane-targeted SuperPLDx30 (magenta) and PA probe GFP-PASS (green). After 30 minutes of blue light illumination, substantial PA accumulates in the plasma membranes, indicated by the PA probe colocalizing with SuperPLDx30. Scale bar 10 µm. Images adapted from Tei et al. 2022, bioRxiv, under a CC-BY-NC-ND 4.0 license.
Just make sure you don’t overdo it! The moderately-enhanced 10x and 30x SuperPLDs avoid dark-state activity and cytotoxicity better than the 50x and 100x variants. But those super-high efficiency constructs are great for in vitro biocatalytic applications where toxicity doesn’t matter. Plus, for folks interested in directed-evolution methods, the activity-based selection in mammalian cells used here could be an attractive strategy for engineering other enzymes.
Conclusion
We hope this post has illuminated some of the many tools available for controlling cell dynamics. Need a refresher on VVDfast, iLID, LOV, CRY2-CIB, and other optogenetic modules? Or want to find more optogenetic tools in our collection? Check out our Optogenetics Guide or browse our collections of optogenetics plasmids – and keep an eye out for future updates to the collection!
Resources and references
Additional resources on the Addgene blog
Viral Vectors 101: Optogenetics
Additional resources on Addgene.org
References
Aljiboury, A. A., Ingram, E., Krishnan, N., Ononiwu, F., Pal, D., Manikas, J., Taveras, C., Hall, N. A., Silva, J. D., Freshour, J., & Hehnly, H. (2023). Rab8, Rab11, and Rab35 coordinate lumen and cilia formation during zebrafish left-right organizer development. PLoS Genetics, 19(5), e1010765. https://doi.org/10.1371/journal.pgen.1010765.
Berlew, E. E., Yamada, K., Kuznetsov, I. A., Rand, E. A., Ochs, C. C., Jaber, Z., Gardner, K. H., & Chow, B. Y. (2022). Designing Single-Component Optogenetic Membrane Recruitment Systems: The Rho-Family GTPase Signaling Toolbox. ACS Synthetic Biology, 11(1), 515–521. https://doi.org/10.1021/acssynbio.1c00604.
Mahlandt, E. K., Palacios Martínez, S., Arts, J. J., Tol, S., van Buul, J. D., & Goedhart, J. (2023). Opto-RhoGEFs, an optimized optogenetic toolbox to reversibly control Rho GTPase activity on a global to subcellular scale, enabling precise control over vascular endothelial barrier strength. eLife, 12, RP84364. https://doi.org/10.7554/eLife.84364.
Meiring, J. C. M., Grigoriev, I., Nijenhuis, W., Kapitein, L. C., & Akhmanova, A. (2022). Opto-katanin, an optogenetic tool for localized, microtubule disassembly. Current Biology, 32(21), 4660-4674.e6. https://doi.org/10.1016/j.cub.2022.09.010.
Ridley, A. J. (2016). Rho GTPase signalling in cell migration. Current Opinion in Cell Biology, 36, 103–112. https://doi.org/10.1016/j.ceb.2015.08.005.
Tei, R., Bagde, S. R., Fromme, J. C., & Baskin, J. M. (2023). Activity-based directed evolution of a membrane editor in mammalian cells. Nature Chemistry, 15(7), 7. https://doi.org/10.1038/s41557-023-01214-0. Preprint on bioRxiv: https://doi.org/10.1101/2022.09.26.509516.
Topics: Optogenetics





Leave a Comment