This post was contributed by guest blogger Jessica Polka, a Postdoctoral Research Fellow with Pamela Silver.
Most types of biological motion (whether endocytosis, vesicle trafficking, or muscle contractions) are produced by orchestrated movements of networks of proteins consuming molecular fuel sources. While the importance of understanding these complex processes can’t be overstated, we can also learn a lot from Nature’s simpler solutions to transmitting forces over long distances. For instance, how much force can be generated by conformational changes in proteins? How can information propagate through a structured material over a long distance? And can we understand such a structure well enough to engineer it to suit our purposes?
In search of simple biological machines
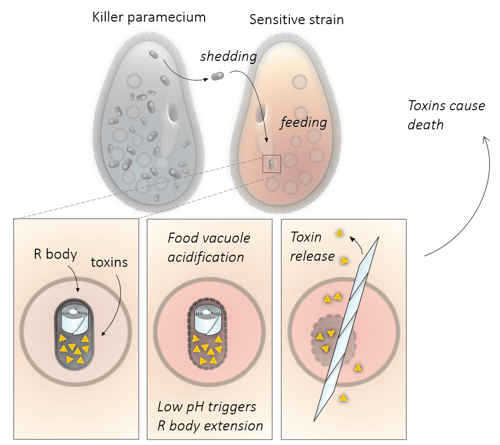
R bodies can repeatedly extend and contract in response to changing pH, and they can rupture E. coli spheroplasts to release encapsulated proteins. Video by ACS. For a more cartoony explanation of R-bodies, check out this video.
 Most types of biological motion (whether endocytosis, vesicle trafficking, or muscle contractions) are produced by orchestrated movements of networks of proteins consuming molecular fuel sources. While the importance of understanding these complex processes can’t be overstated, we can also learn a lot from Nature’s simpler solutions to transmitting forces over long distances. For instance, how much force can be generated by conformational changes in proteins? How can information propagate through a structured material over a long distance? And can we understand such a structure well enough to engineer it to suit our purposes?
Most types of biological motion (whether endocytosis, vesicle trafficking, or muscle contractions) are produced by orchestrated movements of networks of proteins consuming molecular fuel sources. While the importance of understanding these complex processes can’t be overstated, we can also learn a lot from Nature’s simpler solutions to transmitting forces over long distances. For instance, how much force can be generated by conformational changes in proteins? How can information propagate through a structured material over a long distance? And can we understand such a structure well enough to engineer it to suit our purposes?
These questions can be addressed using R bodies: simple, force-generating protein machines that can extend and retract over many microns in response to pH changes without the need for ATP or other chemical fuel sources. Like an actin filament or a microtubule, R bodies are a polymer of many copies of monomeric subunits. Unlike cytoskeletal polymers, however, R bodies do not exert force by dynamically rearranging into different polymerization states. Instead, R body monomers respond to pH changes by undergoing local conformational changes. These tiny movements are then amplified tens or hundreds of thousands of times by propagating through an enormous lattice of identical subunits.
 A brief history of R bodies
A brief history of R bodies
R bodies have been studied for many decades, and they were known to be resistant to detergents and degradation under a variety of conditions. They are composed of 4 proteins encoded in a single operon and were first expressed in E. coli in the 1980s (Kanabrocki et al 1986). Much later, it was shown that putting the operon under a strong promoter results in high R-body expression (Schrallhammer et al 2012). This made them an appealing target for bioengineering, but it wasn’t clear whether they could 1) be decorated with proteins and remain functional, 2) break membranes outside of their natural context or 3) be modified to extend at different pH levels.
Domesticating R bodies
 We have shown that all three of these engineering objectives are possible in our recent paper (Polka and Silver 2016). First, we demonstrated that R bodies decorated with a fluorescent protein remain functional. Attaching the fluorescent protein fusions allowed us to image R bodies inside cells, and these constructs may be useful to future scientists who would like to further study R body dynamics.
We have shown that all three of these engineering objectives are possible in our recent paper (Polka and Silver 2016). First, we demonstrated that R bodies decorated with a fluorescent protein remain functional. Attaching the fluorescent protein fusions allowed us to image R bodies inside cells, and these constructs may be useful to future scientists who would like to further study R body dynamics.
Second, we showed that these fluorescent R bodies can rupture E. coli spheroplasts (cells which have had their cell walls removed with lysozyme). This demonstrates that R bodies can function to break membranes of different organisms.
And finally, we demonstrated that R bodies can be “tuned” with mutagenesis to extend at different pH values. Using a screen that takes advantage of the fact that coiled R-bodies sediment in certain buffers but remain soluble when extended (see figure to the right), we identified R-body mutants that extend at both lower and higher pH. We initially used this screen to find R body mutants that couldn’t extend at a normal pH and later realized that these mutations clustered in a particular region of the R-body. Further mutatgenesis of this region produced R-bodies that could extend at different pHs.
Plasmids expressing expressing both wild type and mutant R bodies can be found at Addgene here.
What can R bodies do for you?
R bodies are robust linear actuators that could be used in a variety of applications. For example, you could use them to lyse cells (and surrounding membranes) in keeping with their natural function (see below). Or you could explore their ability to penetrate other barriers (like vasculature or extracellular matrices) in response to pH changes.
Beyond these applications, purified R bodies could also perform as actuators in microelectromechanical systems (MEMS) devices. They could be functionalized so that they adhere to specific surfaces, or they could be decorated with enzymes for use as a scaffold that changes shape in response to environmental conditions.
More speculatively, R bodies might endow divergent organisms with new functions. For inspiration, we can look to nature: some protists are coated in symbiotic bacteria (called epixenosomes) that contain an R body-like structure. As the bacteria develop, the structure extends to rip the bacteria apart; however, they still remain attached to the ciliate. It’s thought that they function as a kind of armor to protect their host from predation (Petroni et al 2000). With a better understanding of how R bodies assemble and function, we might be able to endow cells with new ways to interact mechanically with their environment.
 Jessica Polka is a Postdoctoral Research Fellow with Pamela Silver in the department of Systems Biology at Harvard Medical School. Follow her on Twitter @jessicapolka.
Jessica Polka is a Postdoctoral Research Fellow with Pamela Silver in the department of Systems Biology at Harvard Medical School. Follow her on Twitter @jessicapolka.
References
1. Kanabrocki, J. A., Quackenbush, R. L. & Pond, F. R. Organization and expression of genetic determinants for synthesis and assembly of type 51 R bodies. J. Bacteriol. 168, 40–48 (1986). PubMed PMID: 3759909. PubMed Central PMCID: PMC213417.
2. Schrallhammer, M. et al. Tracing the role of R-bodies in the killer trait: Absence of toxicity of R-body producing recombinant E. coli on paramecia. European Journal of Protistology 48, 290–296 (2012). PubMed PMID: 22356923.
3. Polka, J. K. & Silver, P. A. A Tunable Protein Piston That Breaks Membranes to Release Encapsulated Cargo. ACS Synth. Biol. (2016). doi:10.1021/acssynbio.5b00237. PubMed PMID: 26814170.
- Find Plasmids from This Publication at Addgene
4. Petroni, G., Spring, S., Schleifer, K.-H., Verni, F. & Rosati, G. Defensive extrusive ectosymbionts of Euplotidium (Ciliophora) that contain microtubule-like structures are bacteria related to Verrucomicrobia. PNAS 97, 1813–1817 (2000). PubMed PMID: 10660683. PubMed Central PMCID: PMC26518.
Additional Resources on the Addgene Blog
- Read About Other Hot Plasmid Technologies
- Read Other Synthetic Biology Posts
- Suggest Plasmid Tools for the Repository
Resources on Addgene.org
- Read our Hot Plasmids Articles
- Find Plasmids for Bacterial Expression
Topics: Synthetic Biology, Other Plasmid Tools, Plasmids






Leave a Comment